Translate this page into:
Overview of Antimicrobial Resistance: An Emerging Silent Pandemic

*Corresponding author: Manita Paneri Department of Medical Microbiology, Centre for Interdisciplinary Biomedical Research, Adesh University, Punjab, Bathinda, India. manitaprashant@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Paneri M, Sevta P. Overview of antimicrobial resistance: an emerging silent pandemic. Glob J Med Pharm Biomed Update 2023;18:11.
Abstract
Before the outbreak of Coronavirus disease-19, one of the top 10 most risks identified by the World Health Organization (WHO) is antimicrobial resistance (AMR) that is also known as “silent pandemic.” According to Lord Jim O’Neill’s report, if no action is taken, then AMR will result in 10 million deaths annually by 2050. In the agricultural and medical sectors, the indiscriminate utilization of antimicrobial agents is getting worse. For the treatment of carbapenem-resistant Gram-negative infections, new antibiotics are urgently required. Microbes, through genetic mutations, acquire resistance to combat with antimicrobial drugs and thus maintain their survival. The WHO on October 25, 2022, released the “Fungal Priority Pathogens List” which includes 19 fungi that pose the highest threat to public health. The implementation of strategies that avoid any possible exposure of pathogens to antibiotics in non-clinical environments involves cooperation between clinicians, researchers, and policymakers. To combat the emerging threat posed by AMR, a multifaceted and holistic approach known as “One health for all” is also required. The curriculum of medical schools needs to be revitalized using a one-health concept. By spreading vital public health information, these initiatives will be successful in promoting awareness among students and the general public.
Keywords
Antimicrobial resistance
Pandemic
Carbapenem resistant
Antibiotic
Biofilm
INTRODUCTION
“Antimicrobial Resistance” (AMR) is a condition in which bacteria, viruses, fungi, and parasites evolve over time and cease to respond to drugs, rendering outbreaks extremely difficult to treat and raising the risk of disease transmission, life-threatening sickness, and death. Each year, more than 700,000 individuals lose their lives due to AMR. The emergence of antibiotic-resistant superbugs, or AMR, has served as a global wake-up call. Before the outbreak of Coronavirus disease-19 (COVID-19), one of the top 10 risks identified by the World Health Organization (WHO) is AMR, which is also known as the “silent pandemic.” The United Kingdom (UK) government has appointed an economist, Lord Jim O’Neill, to conduct a strategic assessment of how to best combat AMR in the UK. According to his report, if no action is taken, then AMR will result in 10 million deaths annually by 2050.[1-3]
The COVID-19 pandemic has highlighted the significance of enhancing hygienic practices as well as infection control, particularly in low- and middle-income nations. Antimicrobials have often been given to patients in the hospital who had COVID-19 symptoms to lessen their risk of developing secondary bacterial infections, which increases the prevalence of resistant strains. Increased usage of disinfectants, such as hand sanitizers and surface cleaners, is projected to result in higher instances of resistant pathogens in the upcoming years.[2,3]
In the agricultural and medical sectors, the indiscriminate utilization of antimicrobial agents (AMAs) is getting worse. Like all other organisms, pathogens evolve by adapting to new environmental factors. When a particular community of microbes is exposed to an antimicrobial, vulnerable pathogens perish while resistant pathogens live. The introduction of new antibiotics in the market was much easier and quicker a century back than it is today. After the initial euphoria, it became apparent that bacteria are capable of developing, acquiring, and disseminating a wide range of resistance mechanisms. As antimicrobial resistance increases internationally, causing diseases that are difficult to treat and ultimately lead to mortality. Antibiotics are becoming less and less effective. For the treatment of carbapenem-resistant Gram-negative infections, new antibiotics are urgently required. AMR reduces productivity by necessitating prolonged hospitalization and thus more expensive, intensive treatment.[3,4]
HISTORY OF ANTIMICROBIAL AGENTS (AMAS)
Bacterial infections were the leading cause of death in the developed world up until the turn of the 20th century. Several civilizations utilized various moulds and plant extracts to cure diseases. In 1910, Paul Ehrlich introduced the arsenic-based synthetic drug “Salvarsan” for treating syphilis caused by Treponema pallidum.[5] Later in 1928, Alexander Fleming discovered “Penicillin.”[6] In 1930, Waksman defined antibiotics as “A compound made by a microbe to destroy other microbes” and Actinomycetes, which inhabit soil, were recognized by him as being active producers of antimicrobial substances. Neomycin and streptomycin, the first antibiotics effective against tuberculosis, were among the several antibiotics produced by soil-dwelling Actinomycetes that Waksman identified. In his ground-breaking research, Waksman identified the genus Streptomyces as a prolific natural product generator.[3,7] The “golden age” of antibiotic discovery, which lasted from the 1940s through the 1960s, was started by Waksman’s efforts but the emergence of AMR has lowered their efficacy. The cephalosporins were initially isolated from the fungus Cephalosporium acremonium cultures by the Italian scientist Giuseppe Brotzu in 1945.[8]
Vancomycin was eventually obtained from Streptomyces orientalis, which became available to patients in 1958.[9] Antibiotic resistance was by that period becoming a problem, therefore researchers looked for novel ways to enhance existing drugs to overcome it. The first penicillinase-resistant β-lactam antibiotic, methicillin, was created by Beecham in 1959. ampicillin was introduced around 1961.[10,11]
As a by-product from Streptomyces clavuligerus culture, beta-lactamase inhibitors were discovered in 1976.[12] These resulted in the development of thienamycin, derived from Streptomyces cattleya in 1976, which was the precursor for the carbapenems, and clavulanic acid, which when coupled with amoxicillin, produced co-amoxiclav.[11,12] Around 1987, a new class of antimicrobials had been discovered and introduced to the market. Since then, there has not been much advancement in this area, and there are not many novel antimicrobial classes in the pipeline right now. The paucity of antibiotics for Gram-negative bacteria is particularly concerning. The timeline of the major antibiotics’ discoveries and the mode of action is depicted in [Figures 1 and 2].
![Timeline of antimicrobials discovery.[3]](/content/119/2023/18/1/img/GJMPBU-18-11-g001.png)
- Timeline of antimicrobials discovery.[3]
![Antimicrobials classification on the basis of mode action.[3]](/content/119/2023/18/1/img/GJMPBU-18-11-g002.png)
- Antimicrobials classification on the basis of mode action.[3]
MICROORGANISMS INVOLVED IN AMR
Microbes through genetic mutations acquire resistance to combat with antibiotics and thus maintain their survival. When in 1941 “Penicillin” was first commercially available in the market then Streptococci, Staphylococci, and Gonococci were apparently found to be resistant first; then in 1942, Staphylococcus aureus was found to be resistant to penicillin and later in 1960, it was found resistant to methicillin.[3] Later on, other microbes gradually developed resistance to various antibiotics including carbapenem which is considered as a last resort to treat gram-negative infections.[13] The WHO in 2017 published a list of high-priority pathogens that were divided into high, medium, and critical priority[14,15] [Table 1].
| Critical | High | Medium |
|---|---|---|
| Acinetobacter baumannii | Enterococcus faecium | Streptococcus pneumoniae |
| Pseudomonas aeruginosa | Staphylococcus aureus | Haemophilus influenzae |
| Enterobacteriaceae | Helicobacter pylori | Shigella spp. |
| Campylobacter spp. | ||
| Salmonellae | ||
| Neisseria gonorrhoeae |
The WHO is more concerned about “ESKAPE” (“Enterococcus faecium, S. aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter spp.”) pathogens as they have emerged as multidrug resistance (MDR), pan drug resistance and extensively drug-resistant, infections caused by them are very difficult to treat.[16]
The diseases caused by fungi have been neglected for so long that we barely comprehend the extent of the problem. The rising problem of invasive fungal diseases prompted the establishment of the “Fungal Priority Pathogens List” by the WHO on October 25, 2022, which includes 19 fungi that pose the highest threat to public health. Patients who are critically unwell and those with serious underlying immune system-related disorders usually have these fungus infections in their invasive forms[17] [Table 2].
| Critical | High | Medium |
|---|---|---|
| Cryptococcus neoformans | Candida glabrata | Scedosporium spp. |
| Candida auris | Histoplasma spp. | Lomentospora prolificans |
| Aspergillus fumigatus | Eumycetoma causative agents | Coccidioides spp. |
| Candida albicans | Mucorales | Candida krusei |
| Fusarium spp. | Cryptococcus gattii | |
| Candida tropicalis | Talaromyces marneffei | |
| Candida parapsilosis | Pneumocystis jirovecii | |
| Paracoccidiodes spp. |
Human immunodeficiency virus (HIV) genetic variations limit the capacity of medications to stop the virus from replicating, which is the cause of HIV treatment resistance. Due to the advent of drug-resistant viruses, all antiretroviral drugs, even those belonging to more recent pharmacological classes, run the possibility of becoming partially or completely inactive. The emergence and dissemination of drug resistance, notably to artemisinin and partners on Artemisinin combination therapies, the first-line treatment advised by the WHO, pose a danger to the control and eradication of malaria. Three of the five malaria species that afflict humans, Plasmodium vivax, Plasmodium falciparum, and Plasmodium malariae have been discovered to be resistance to antimalarial drugs.[18]
RESISTANT MECHANISM
AMR has evolved to be a major cause of morbidity and mortality throughout the world. It is anticipated that overall awareness of such strategies eventually results in more effective treatments for infectious diseases.[19] There are various strategies such as intrinsic resistance, horizontal gene transfer, biofilm formation and mutations etc. through which microbes have adopted resistance against antimicrobial medicines [Figure 3].

- Resistance mechanism of bacteria.
Following are the methods through which microbes acquired resistance to antimicrobial drugs:
Porin mediated
The plasma-membrane of Gram-positive bacteria is encased in a hard, stiff mesh known as the “cell wall.” Gram-negative bacteria have a thin cell wall and an outer membrane, which is a second lipid membrane. The outer membrane comprises proteinaceous porin channels that permit the entry of many substances, including antibiotics.[20] Choi and Lee (2019) in their research on Escherichia coli’s outer membrane porins found that OmpF mutant was resistant to several antibiotics including β-lactams.[21] Liu et al. (2022) found that the deletion of the hopE and hopD porin genes lowered the amount of streptomycin in the Helicobacter pylori and, hence, restored the expression of the resistant gene, which was suppressed by streptomycin in the wild-type strain.[22] A study of the outer membrane proteins of E. coli and K. pneumoniae isolates done by Khalifa et al. (2021), revealed that 93.3% and 95.7%, respectively, had porins that had been deleted or altered. In addition, frameshift mutations in some isolates’ porin genes were also found by sequence analysis.[23]
Overexpression of efflux pump
The transporter protein referred to as “efflux pumps” is found in bacterial cells and is located in the cytoplasmic membrane. These are required to evacuate various drugs, dyes, and detergents. Mutations in the efflux pump system cause these pumps to be overexpressed, which minimizes the accumulation of antibiotics in the bacterial cell and, thus, helps bacteria get rid of these antibiotics.[24] According to Pandey et al. (2020), fluconazole resistance in Candida tropicalis, which is responsible for approximately 40% of candidemia, is acquired by the overexpression of efflux pump transporter genes and ERG11 mutations.[25]
In the K. pneumoniae strain (KPN142), which is pan-drug resistant, the overexpression of phoPQ causes the resistance to colistin B, and the overexpression of the AcrAB-TolC efflux pump facilitates the resistance to the majority of conventional medical antimicrobials, according to Lv et al., 2021.[26]
Bankan et al. (2021) in their research study found that 19 out of the 42 A. baumannii isolates that were tigecycline-resistant exhibited efflux pump activity. The adeB gene was expressed by all 19 strains.[27]
Plasmids with antibiotic resistance genes
“Plasmids” are extra-chromosomal double-stranded deoxyribonucleic acid (DNA) within a bacterial cell and replicate independently. Plasmids contain various antibiotic-resistance genes that are beneficial to bacteria for their survival. These plasmids are capable of transfer resistance genes through horizontal gene transfer and during conjugation. Cross-species transfer within different groups of bacteria is also possible due to plasmids. Plasmids are ideal vectors for the spread of AMR as they are capable of acquiring novel genes through mobile genetic elements such as transposons or insertion sequences.[28]
Oxacillinase-encoding genes (OXA-23, OXA-24, and OXA-58) and their variants that confer carbapenem drug resistance are acquired by A. baumannii through plasmids. ColE1-type replication is used by K. pneumoniae plasmids that are 25 kb or less in size. Many of these plasmids contained the transposon Tn1331 and its variants.[29] Given the scarcity of available antibiotics to treatment, hospital-associated infections are frequently plagued by resistance to third-generation cephalosporins, which is frequently caused by extended-spectrum beta-lactamases (ESBLs). The ESBL genes are frequently found on large plasmids that move horizontally between Enterobacteriaceae isolates and species. Hawkey et al., 2022 in their research study, sequenced the genome of K. pneumoniae isolates and identified 25 distinct plasmids in which plasmid A was found to be carried blaCTX-M-15 resistant gene. It caused 50% of all ESBL occurrences during the 1st year of their study and was spread at least 4 times into various Klebsiella spp.[30]
Mutation
When microbes are exposed to higher concentrations of antimicrobials, then natural selection plays a role and mutations occur in the genome that gives rise to resistant pathogens, while wild-type strains are eliminated from the environment. These resistant variants can transfer mutant genes to other microbes naturally during conjugation or through spontaneous mutation and may provide intrinsic resistance. For example, A. baumannii has the OXA-51 intrinsic resistant gene that is only specific for it.[31]
In their research on Elezabethkingia anophelis, Lin et al. (2018) observed that out of 67 isolates, 11 isolates showed mutations in DNA gyrase subunit A that indicated high levels of fluoroquinolone resistance.[32] Bachmann et al. (2020), through Sanger sequencing, found mutations at the 23S ribosomal ribonucleic acid (rRNA) locus (macrolide resistance-associated mutations), as well as in the parC and gyrA genes (quinolone-associated mutations) of Mycoplasma genitalium that causes urethritis.[33] According to Nagy et al., 2022 the gmhD genetic variant of Shigella sonnei (strain 4351) caused an enhanced resistance to the cephalosporin and macrolide. This is due to frameshift mutation in the genome of S. sonnei.[34]
Antibiotic-modifying enzyme or Antibiotic degrading enzyme
A vast number of enzymes inactivate antimicrobials by modifying or degrading their structure. All penicillin-binding proteins (PBPs) are targeted by β-lactam antibiotics at their C-terminus [Figure 4].

- Antibiotic resistance due to mutations in penicillin-binding protein.
Gram-positive bacteria develop resistance to β-lactam antibiotics as a result of mutations in PBPs. PBP2a is a mutant form that is encoded by the MecA gene and exhibits only transpeptidase activity, thus conferring resistance to S. aureus against methicillin.[35] The methylation of the 16S rRNA A-site by bacterial 16S rRNA methyltransferases, which impairs the potential of antimicrobials to bind to the ribosome, provides resistance to aminoglycosides.[36]
Antibiotic modifying enzymes can be divided in three parts –
Hydrolases – Esterases, β-lactamases, Epoxide hydrolases
Transferases – Acetyltransferases, Phosphotransferases, Glycosyltransferases, Neucleotidyltransferases, etc.
Redox enzymes – Monooxygenases, Lyases [Figure 5].
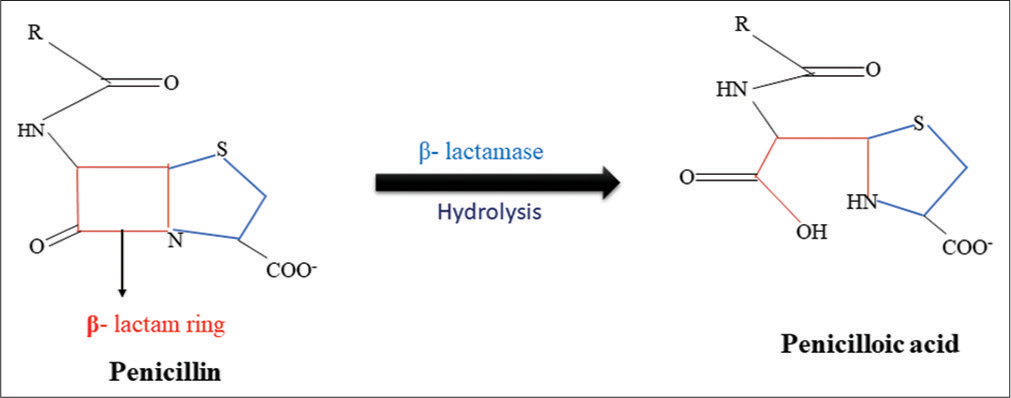
- Hydrolysis of β-lactam ring by β-lactamase.
β-lactam antibiotics have amide bond in the β-lactam ring which is hydrolyzed by β-lactamases. Esterases are responsible for the degradation of the lactone ring that leads to resistance against 14- and 15-membered macrolides. Bacteria are mainly resistant to chloramphenicol due to the production of chloramphenicol acetyltransferases. These enzymes catalyze the addition of the acetyl group of acetyl-CoA to the 3-hydroxyl group of chloramphenicol or its synthetic counterparts (Azidamphenicol and Thiamphenicol), which prevents adhering to ribosomes. The 2’-OH group of the macrolide ring is glycosylated by macrolide glycosyltransferases that causes inactivation of macrolides. By modifying the hydroxyl group, several enzymes deactivate rifamycin. Several tetracyclines, and tigecycline, are resistive to monooxygenase.[37]
Biofilm formation
Bacteria shield themselves from antibiotics, disinfectants, and host inflammatory responses by establishing a biofilm. Biofilm is a complex microbial structure that attaches to a surface and includes a wide range of microbes. Especially in nosocomial conditions, it is among the major causes of infection recurrence [Figure 6].[38]

- Biofilm formation by microbes.
Antimicrobial compounds are significantly less effective against microorganisms in biofilms than they are against planktonic forms.[39] Both Gram-positive bacteria such as S. aureus, Staphylococcus epidermidis, Streptococcus pneumoniae, and Gram-negative bacteria such as P. aeruginosa, K. pneumoniae, A. baumannii, and E. coli, are capable to form biofilm.[40]
Quorum sensing (QS) mechanisms are used by microbes to interact with one another within biofilms. These systems rely on chemical signals. Communication affects density-based pathophysiology, cellular processes, genetic material transfers between cells, dietary uptake, motility, and the production of secondary metabolites. Oligopeptides are used by the Gram-positive bacteria as a signaling molecule whereas Gram-negative bacteria utilize homoserine lactone dependent QS system.[41]
Dimitrova et al., 2021 in their research work on swine feces and lagoons found 16 E. coli strains, of which 31.2% exhibited strong biofilm formation, 37.5% formed moderate biofilm whereas 25% formed weak biofilm and 87.5% were MDR.[42] Biofilm of K. pneumoniae can promote colonization in the gastrointestinal, urinary, and respiratory tracts as well as the emergence of nosocomial infections such as ventilator associated pneumonia in immunocompromised patients.[43] It is challenging to anticipate which compounds may develop into novel therapeutic antifungal drugs as Candida albicans biofilms are still not very susceptible to therapeutic agents already in use.[44]
OTHER CAUSES OF AMR
Human waste
The resistant strains develop when unmetabolized or partially digested pharmaceuticals are discharged into health-care waste. Chaudhary and Uddin, 2022, found cefixime-resistant bacteria in the effluents of Chittagong Medical College Hospital, Bangladesh.[45] Antibiotics, antifungals, and pathogens can be found in feces. The sewage treatment plants aren’t really designed primarily to get rid of them, and due to overflow, leakage, or untreated wastewater, contamination ultimately results in various diseases and the proliferation of resistant strains might happen.[46,47]
Agriculture industry
In the agriculture industry, antibiotics are used to prevent crop diseases and enhance the yield of crops. According to Miller et al., 2022 phytopathogens are controlled by the application of fungicides, antibacterial drugs, and other pesticides. These pathogens have become more resistant to oxytetracycline, copper-based compounds, streptomycin, and some fungicides as a consequence of their application. In Europe, a recent increase has been seen in the prevalence of Aspergillus fumigatus, which causes human aspergillosis and is resistant to triazole.[48,49]
One of the most frequently used chemicals in agriculture is pesticide. According to Liao et al., 2021 three commonly used herbicides, that is, glufosinate, glyphosate, and dicamba enhanced the incidence of mobile genetic elements and AMR genes in soil microbiomes. This might exacerbate the issue of worldwide antibiotic resistance in agricultural ecosystems.[50]
Aquaculture industry
Aquaculture aids in food security and improves the state of the world economy. Untreated or inadequately managed waste and effluents from many sources are disposed of carelessly, allowing numerous pollutants, such as bioactive substances and unmetabolized antibiotics, to enter the environment.[51]
Antimicrobials such as aminoglycosides, macrolides, quinolones, tetracycline, and sulphonamides are used in aquaculture systems for both treatment and prevention. As a consequence, the microbes in the vicinity developed AMR, and other microbes can become resistant through horizontal gene transfer, allowing these resistant microbes to enter humans through the food chain and cause disease. Approximately 80% of the medicines ingested are not absorbed and end up in the surroundings through feces. After being absorbed, antimicrobials are then eliminated by the urine and other secretions and accumulate in the sediments, increasing their concentration in the aquarium, which causes resistance as microbes are now under selection pressure for survival.[52]
Animal husbandry
In animal husbandry, AMAs are used to treat various diseases as well as act as growth promoters.[53] Developing countries adopted intensive farming due to the high demand for animal proteins. Resistant pathogens that are associated with animals are a great threat to humans as they can be easily transmitted from animals to humans through the food chain and are widely distributed in the environment. Inadequate government policies, as well as the limited adoption of infection control strategies, all contribute to the continued use of non-essential antimicrobials in animal husbandry.[54]
Salmonella are zoonotic microorganisms that exhibit antibiotic resistance, particularly to drugs recovered from farm animals. Salmonella can be found in the native gut flora of chickens, hence makes them a reservoir of these bacteria. Animal feed is manufactured in underdeveloped nations in an unregulated way without veterinary oversight or microbiological quality testing. Furthermore, some feed producers include antibiotics in poultry feeds to ward off diseases and flock attrition. These procedures might promote antibiotic-resistant pathogens in the intestinal flora of chickens.[55] Several antibiotics used in veterinary medicine possess the same mode of action as those used to combat bacterial infections in humans, and if not fully metabolized, animal manure will become a source of antibiotic contamination and reservoir for MDR pathogens in the soil.[56] According to Wu et al., 2022, sewage sludge, chicken, swine, and cattle manure are the major sources of contamination for sulphonamides, tetracyclines, fluoroquinolones, and associated ARGs in agricultural soils.[57]
Overuse and misuse of antibiotics
According to Jani et al., 2021, genomic variability as well as the indiscriminate use of pharmaceuticals enable microbes to adjust to the effects of antimicrobials, resulting in a sharp rise in AMR. The reasons behind the increase in treatment failure and the expansion of AMR include self-medication, the availability of antibiotics over the counter, and the prescription of broad-spectrum antibiotics.[58]
Several people frequently use medicines without a prescription, and if they are successful in overcoming a specific ailment, they may employ the same medications to treat other diseases in the future. They may also advise others to do the same. Many folks skip follow-up appointments and do not finish their prescriptions as their doctors have advised.
STRATEGIES TO OVERCOME AMR
The CDC has granted $22 million to 28 organizations across the globe to start two new networks that will combat health-care-associated infections and antimicrobial resistance.[59] The implementation of strategies that avoid any possible exposure of pathogens to antibiotics in non-clinical environments involves cooperation between clinicians, researchers, and policymakers. To combat pathogenic microbes, it is indeed clinically important to use novel and quasi therapies, which are becoming more prevalent in pathogenic bacteria due to increased AMR[60] [Table 3].
| S. No. | Strategies against resistance mechanism | References |
|---|---|---|
| 1. | Combination drug therapy | Bianco et al., 2022; Henson et al., 2016; Li et al., 2019 |
| 2. | CRISPR technology | Rodrigues et al., 2019; Dong et al., 2018 |
| 3. | Nano-drugs delivery | Kang et al., 2017; Bruna et al., 2021 |
| 4. | Bacteriophage therapy | Yazdi et al., 2019; Yang et al., 2020; Roach et al., 2017 |
| 5. | Vaccines | Kaufhold et al., 2019; Barchitta et al., 2022 |
| 6. | Antimicrobials peptides | Liu et al., 2018 |
| 7. | Faecal microbiota transplantation | Freedman et al., 2014; Ueckermann et al., 2020; Bilinski et al., 2017 |
| 8. | Use of Probiotics and Prebiotics | Buyukeren et al., 2020 |
| 9. | Antimicrobial stewardship practices | World Health Organization, 2015 |
| 10. | Global Surveillance approaches | World Health Organization, 2015 |
| 11. | Awareness and Education | Marvasi et al., 2021 |
Combination drug therapy
Combination therapy has been developed by specialists for use against pathogens especially MDR microbes. Combination therapy requires the integration of two or more pharmaceuticals to boost or enhance each one’s efficacy.[61] Carbapenem drugs are considered a last resort to treat Gram-negative infections, but global resistance to these drugs is a worrisome situation as pathogens produce lactamase enzyme as a defense mechanism. Bianco et al., 2022, reported that when avibactam, vaborbactam, and relebactam were combined separately with cefiderocol, the minimal inhibitory concentration (MICs) of cefiderocol showed a 4–256 reduction against carbapenem resistant K. pneumoniae, which was harboring virulent Klebsiella pneumoniae carbapenemase (KPC)-resistant genes. They demonstrated that cefiderocol MICs of K. pneumoniae strains that were harboring blaKPC-41, blaKPC-31, blaKPC-53, and blaKPC-66 genes dropped by 4–64-fold in response to tazobactam.[62]
Another study has been done by Henson et al. (2016) to investigate synergistic effect of daptomycin in combination with piperacillin-tazobactam and ampicillin-sulbactam against methicillin resistant S. aureus and found that six out of eight Methicillin-resistant Staphylococci strains showed synergistic effect.[63] Li et al. (2019) in their study mentioned that when D-penicillamine is combined with fluconazole then it showed synergistic effect against C. albicans as well as against its biofilms that formed within 12 h in vitro.[64]
CRISPR technology
It is becoming more challenging for us to tackle infectious diseases and generate new pharmaceuticals as a result of the emergence of antibiotic-resistant microbes. Innovative drugs that can kill MDR pathogens and distinguish between beneficial and dangerous species may be developed utilizing CRISPR-Cas technology.[65] CRISPR-Cas9 is also known as “RNA-guided-DNA cutter.” The Cas system encodes minute phage genome sequences into the bacterial genome to launch a defense after bacteriophage invasion. Given that Cas9 has nuclease activity, it is indeed possible to program it to produce a highly specific sequence.[66]
Target specificity, which enables differentiation between commensal and harmful microorganisms, is an important feature of CRISPR/Cas. Guide RNAs can be designed to specifically target pathogen-specific genes, antibiotic resistance genes, and virulence genes. This ensures that innocuous strains are maintained while pathogenic variants are terminated in a species-specific approach. According to research conducted by Rodrigues et al., 2019, the presence of antibiotic-resistant E. faecalis is reduced significantly by the administration of CRISPR-Cas antimicrobial drugs inside the gut of mice. They revealed that E. faecalis donor strains carrying CRISPR-Cas antimicrobials are resistant to in vivo acquisition of resistance genes.[67]
In their research study, Dong et al. (2018), created a host-independent conjugative plasmid and employed an engineered CRISPR/Cas9 system to eliminate bacteria carrying the mCR-1 plasmid. They discovered that conjugative plasmids can cause the recipient cell to develop resistance against the mCR-1-interacting plasmid in addition to serving as a novel tool for eliminating resistant plasmids and making the target bacterium more susceptible to antibiotics.[68]
Nanodrugs delivery
In order to develop effective therapeutic approaches for MDR planktonic pathogens that form biofilms, nanotechnology offers a new set of tools. Nanoparticles are able to circumvent current resistance mechanisms and might be less likely to promote resistance than traditional antibiotics.[69] Their exceptionally small size and high surface-to-volume ratio enable their unique efficacy in therapeutics. In the treatment of diseases spurred on by intracellular pathogens and MDR isolates, this provides a significant competitive advantage over traditional medications.[70] By effectively delivering the Cr-Nanocomplex to Methicillin-resistant S. aureus, Kang et al., 2017, showed that it can be even more effective at editing the bacterial genome than native Cas9 complexes or traditional lipid-based formulations.[71] According to Bruna et al., 2021, silver nanoparticles (AgNPs) have been promoted as a superior antibacterial agent capable of battling microbes that cause disease both in vitro and in vivo. AgNPs have several, concurrent modes of action, and they have a synergistic effect when combined with antimicrobials to combat pathogens like E. coli and S. aureus.[72]
Bacteriophage therapy
Utilizing viruses, phage therapy (PT) tackles bacterial infection. Phages or bacteriophages are the terms for bacterial viruses. PT primarily uses obligately lytic phage to eliminate the associated microbial host while sparing human cells and minimizing the major influence on commensal bacteria that frequently arises from antibiotic use.[73]
“Staphylococcus saprophyticus” is a Gram-positive, nonhemolytic, and catalase-positive bacteria that causes urinary tract infections, especially in females. Yazdi et al., 2019, isolated lytic phage (vB_SsapS-104) against S. saprophyticus and found that it was able to lyse eight out of the nine clinical isolates in vitro received from a hospital in Gorgan, Iran.[74]
Yang et al., 2020 isolated virulent Enterococcus faecalis bacteriophage PHB08. They demonstrated that when used against E. faecalis biofilms, PHB08 and its endolysin lys08 both exhibited antibiofilm activity.[75] According to Roach et al., 2017, the host immune system and bacteriophage should work synergistically for PT to really be effective against an acute respiratory pathogen.[76]
Vaccines
Vaccines are administered as a preventative measure and are effective before bacteria commence to proliferate after the onset of infection. This significantly lowers the risk that mutation conferring resistance will arise and propagate.[77]
According to Kaufhold et al., 2019, typhoid conjugate vaccines (TCVs) have potential to treat diseases caused by antimicrobial resistant Salmonella typhi.[78] The WHO-recommended TCV was incorporated to Pakistan’s routine immunization schedule on November 15, 2019, making it the first nation in the world to do so.[79] Barchitta et al., 2022, demonstrated that Influenza vaccination coverage in patients over 64 and AMR in strains of E. coli and K. pneumoniae are significantly inversely correlated.[80] Vaccines stimulate the immune system to locate pathogens and mount an immediate and robust immunological counterattack. A phenomenon known as herd immunity allows numerous vaccines to safeguard the population’s unvaccinated individuals who are unable to receive vaccinations. Some effective vaccines also inhibit pathogen colonization in patients, which leads to highly effective herd immunity.[81]
Antimicrobial peptides (AMPs)
Tiny peptide known as AMPs is abundantly found in nature. AMPs can suppress a wide variety of bacteria, fungi, parasites, and viruses. They have a promising future in the fields of medicine, food, agriculture, animal husbandry, and aquaculture.[82]
In eukaryotes, AMPs play a significant role in the innate immune system. They are formed by host cells as part of long-term immune surveillance against pathogen invasion. Numerous AMPs have been identified in a wide range of taxa, including microorganisms, amphibians, fish, reptiles, mammals, birds, and invertebrates since the very first AMP was discovered in the chrysalis of the American silkworm. Nisin, which is synthesized by the lactic Streptococci spp. was the first AMP identified in bacteria, and is poisonous to other bacterial species. Copsin, which is derived from the mushroom Coprinopsis cinerea, has bactericidal properties toward certain Gram-positive bacteria. In addition, the intestinal microbiota generated AMPs.[83] AMPs have the potential to penetrate cell membranes, inhibit DNA replication and protein synthesis, and disrupt cell division. Defensins, one type of AMP, can stimulate proinflammatory cytokines like interleukin-1. Pseudomonas aeruginosa-induced pneumonia in mice was already improved by cathelicidin BF’s immunomodulatory effect, according to research done by Liu et al., 2018.[84,85]
Fecal microbiota transplantation (FMT)
Antibiotic resistant microbes (AROs) may accumulate in the gastrointestinal system and remain there without manifesting any pathological changes. ARO infection and ARO transmission to other individuals are threats for patients who already have ARO colonization.[86] Critically ill patients who are prescribed broad-spectrum antibiotics and several types of organ support often have dysbiosis of the gut microbiota.[87]
In 2014, Freedman and Eppes claimed that they were the first to use FMT to eliminate the colonization of K. pneumoniae that was harboring virulent KPC genes. They suggested that patients suffering from resistant enteric pathogens should be treated by FMT.[88]
Ueckermann et al., 2020 reported a case of critically ill patient who was suffering from recurrent MDR K. pneumoniae and successfully treated with FMT. The donor’s gut microbiome’s 16S rRNA sequencing revealed a preponderance of Bacteroides and Firmicutes whereas Proteobacteria were more prevalent in the patient.[89] According to Bilinski et al., 2017 FMT is sage and effective therapy for patients having blood disorder.[90]
Use of probiotics and prebiotics
“Probiotics” are viable, live, non-pathogenic bacteria (i.e., Lactobacillus spp., Bifidobacterium spp. etc), yeast (i.e., Saccharomyces boulardii), and other microbes that improve human health whereas “prebiotics” are non-digestible substances that commensal microbes inside the human gut preferentially degrade. They foster a hospitable environment for commensal proliferation and boost microbial diversity, enhancing human health. Prebiotics can be found in lactulose, inulin, glucose-, fructose-, and xylo-oligosaccharides.[91-93]
Buyukeren et al., 2020 reported that vancomycin-resistant Enterococcus (VRE) is cleared more quickly in new-born patients who utilized Lactobacillus rhamnosus GG. In their research study, they demonstrated that 21 out of 22 patients got rid of VRE within 6 months.[94]
Antimicrobial stewardship practices (AMSPs)
In 2015, the WHO established a global action plan that requires its member states to develop national strategy for AMR. The term “AMSP” refers to comprehensive plans created for the appropriate use of AMAs using best antimicrobial drug, dosage, treatment regimen, and administration method with the least amount of toxicity.[95]
The goal for AMSPs is to encourage, enhance, oversee, and assess the prudent application of antimicrobial drugs. Critical initiatives toward preventing and minimizing AMR involve establishing AMSP policies, principles, training healthcare professionals, and providing effective interventions. To combat the emerging threat posed by AMR, a multifaceted and holistic approach known as “One Health for All” is also required. The usage of antimicrobials has been reduced by 22–36% as a result of (AMSP), as well as considerable financial benefits.[96]
Global surveillance approaches
Global Antimicrobial Resistance and Use Surveillance system (GLASS) was introduced in 2015 by the WHO. GLASS promotes nations to switch from relying solely on laboratory data to surveillance methods based on systems that incorporate epidemiological, clinical, and population-level data. 127 nations, territories, and regions are going to be part of GLASS by the ending of 2022.[97,98]
Awareness and education
“World antimicrobial awareness week” is celebrated every year on 18–24 November across the world. Many activities such as theatre play, music, poster making, paper presentation, debate, story-telling, quiz etc. have been suggested to involve students and the general public in education programs in the context of the WHOs One-Health initiative. By spreading vital public health information, these initiatives are successful in promoting awareness among students and the general public. The curriculum of medical schools needs to be revitalized using a One-Health concept. High schools and undergraduate courses must adopt the integration of discovery and research-based methodologies. Fostering women’s inclusion in the fields of science, technology, engineering, and mathematics is an essential strategy for influencing and engaging more individuals from various societies and social backgrounds.[99]
CONCLUSION
AMR is a great threat to all flora and fauna. It cannot be solved by a single person, group, organization, or industry. A multisectoral and collaborative environment is required to tackle the complicated problem of AMR. The “One Health” strategy brings together a wide range of stakeholders and sectors. For discovering long-lasting remedies, more interventions are required. Global surveillance systems, antibiotic stewardship practices, and awareness and education for a judicious use of antimicrobials are need of the hour to tackle AMR at the global level.
Acknowledgement
The authors are thankful to the Chairperson Prof. R.G. Saini, Centre for Interdisciplinary Biomedical Research, Dean, Post Graduate Studies & Research, Adesh University, for his motivation and support.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Antibiotic resistance: Calling time on the 'silent pandemic' JAC Antimicrob Resist. 2022;4:dlac016.
- [CrossRef] [PubMed] [Google Scholar]
- Antimicrobial resistance and the COVID-19 pandemic. Bull World Health Organ. 2022;100:295-295.A.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotics: Past, present and future. Curr Opin Microbiol. 2019;51:72-80.
- [CrossRef] [PubMed] [Google Scholar]
- Increased antimicrobial resistance during the COVID-19 pandemic. Int J Antimicrob Agents. 2021;57:106324.
- [CrossRef] [PubMed] [Google Scholar]
- Organometallic compounds in drug discovery: Past, present and future. Drug Discov Today Technol. 2020;37:117-24.
- [CrossRef] [PubMed] [Google Scholar]
- The Conquest of Tuberculosis California, United States: University of California Press; 2021.
- [Google Scholar]
- Editors of Encyclopaedia: Cephalosporin In: Encyclopedia Britannica. Available from: https://www.britannica.com/science/cephalosporin [Last accessed on 2022 Sep 27]
- [Google Scholar]
- An overview of analytical methodologies for determination of vancomycin in human plasma. Molecules. 2022;27:7319.
- [CrossRef] [PubMed] [Google Scholar]
- Current challenges and advancements towards discovery and resistance of antibiotics. J Mol Struct. 2022;1248:131380.
- [CrossRef] [Google Scholar]
- Orally administered amoxicillin/clavulanate: Current role in outpatient therapy. Infect Dis Ther. 2021;10:15-25.
- [CrossRef] [PubMed] [Google Scholar]
- Rational selection of culture medium for clavulanic acid production by Streptomyces clavuligerus based on a metabolic modeling approach. Agric Nat Resour. 2022;56:267-76.
- [CrossRef] [Google Scholar]
- Antibiotics: From prehistory to the present day. J Antimicrob Chemother. 2016;71:572-5.
- [CrossRef] [PubMed] [Google Scholar]
- WHO Global Priority Pathogens List: A bibliometric analysis of medlinepubmed for knowledge mobilization to infection prevention and control practices in Bahrain. Oman Med J. 2019;34:184-93.
- [CrossRef] [PubMed] [Google Scholar]
- Available from: https://www.who.int/news/item/27-02-2017-who-publishes-list-of-bacteria-for-which-new-antibiotics-are-urgently-needed [Last accessed on 2022 Dec 18]
- Emerging strategies to combat ESKAPE pathogens in the era of antimicrobial resistance: A review. Front Microbiol. 2019;10:539.
- [CrossRef] [PubMed] [Google Scholar]
- Available from: https://www.who.int/news/item/25-10-2022-who-releases-first-ever-list-of-health-threatening-fungi [Last accessed on 2022 Nov 10]
- Available from: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance [Last accessed on 2022 Nov 10]
- An overview of the antimicrobial resistance mechanisms of bacteria. AIMS Microbiol. 2018;4:482-501.
- [CrossRef] [PubMed] [Google Scholar]
- Action and resistance mechanisms of antibiotics: A guide for clinicians. J Anaesthesiol Clin Pharmacol. 2017;33:300-5.
- [CrossRef] [PubMed] [Google Scholar]
- Distinct roles of outer membrane porins in antibiotic resistance and membrane integrity in Escherichia coli. Front Microbiol. 2019;10:953.
- [CrossRef] [PubMed] [Google Scholar]
- HopE and HopD porin-mediated drug influx contributes to intrinsic antimicrobial susceptibility and inhibits streptomycin resistance acquisition by natural transformation in Helicobacter pylori. Microbiol Spectr. 2022;10:e0198721.
- [CrossRef] [PubMed] [Google Scholar]
- β-lactam resistance associated with β-lactamase production and porin alteration in clinical isolates of E. coli and K. pneumoniae. PLoS One. 2021;16:e0251594.
- [CrossRef] [PubMed] [Google Scholar]
- Detection of overexpression of efflux pump expression in fluoroquinolone-resistant Pseudomonas aeruginosa isolates. Int J Appl Basic Med Res. 2020;10:37-42.
- [CrossRef] [PubMed] [Google Scholar]
- Overexpression of efflux pump transporter genes and mutations in ERG11 pave the way to fluconazole resistance in Candida tropicalis: A study from a North India region. J Glob Antimicrob Resist. 2020;22:374-8.
- [CrossRef] [PubMed] [Google Scholar]
- Overexpression of efflux pumps mediate pan resistance of Klebsiella pneumoniae sequence Type 11. Microb Drug Resist. 2021;27:1405-11.
- [CrossRef] [PubMed] [Google Scholar]
- Overexpression of the adeB efflux pump gene in tigecycline-resistant Acinetobacter baumannii clinical isolates and its inhibition by (+) usnic acid as an adjuvant. Antibiotics (Basel). 2021;10:1037.
- [CrossRef] [PubMed] [Google Scholar]
- Plasmids carrying antimicrobial resistance genes in Enterobacteriaceae. J Antimicrob Chemother. 2018;73:1121-37.
- [CrossRef] [PubMed] [Google Scholar]
- Small Klebsiella pneumoniae plasmids: Neglected contributors to antibiotic resistance. Front Microbiol. 2019;10:2182.
- [CrossRef] [PubMed] [Google Scholar]
- ESBL plasmids in Klebsiella pneumoniae: Diversity, transmission and contribution to infection burden in the hospital setting. Genome Med. 2022;14:97.
- [CrossRef] [PubMed] [Google Scholar]
- Evolutionary pathways and trajectories in antibiotic resistance. Clin Microbiol Rev. 2021;34:e00050-19.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical manifestations, molecular characteristics, antimicrobial susceptibility patterns and contributions of target gene mutation to fluoroquinolone resistance in Elizabethkingia anophelis. J Antimicrob Chemother. 2018;73:2497-502.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of Mycoplasma genitalium infection, antimicrobial resistance mutations, and symptom resolution following treatment of urethritis. Clin Infect Dis. 2020;71:e624-32.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of mutation in lipopolysaccharide biosynthesis on bacterial fitness. Cells. 2022;11:3249.
- [CrossRef] [PubMed] [Google Scholar]
- Aminoglycoside resistance: The emergence of acquired 16S ribosomal RNA methyltransferases. Infect Dis Clin North Am. 2016;30:523-37.
- [CrossRef] [PubMed] [Google Scholar]
- Rox, a rifamycin resistance enzyme with an unprecedented mechanism of action. Cell Chem Biol. 2018;25:403-12.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotics versus biofilm: An emerging battleground in microbial communities. Antimicrob Resist Infect Control. 2019;8:76.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotic resistance in biofilm In: Bacterial Biofilms. London: IntechOpen; 2020.
- [CrossRef] [Google Scholar]
- Introductory chapter: Understanding infections caused by opportunistic bacterial pathogens In: Pseudomonas aeruginosa-Biofilm Formation. Infections and Treatments. London: IntechOpen; 2021.
- [CrossRef] [Google Scholar]
- Communication is the key: Biofilms, quorum sensing, formation and prevention. Discoveries (Craiova). 2019;7:e100.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of antibiotic-resistant Escherichia coli isolated from swine faeces and lagoons in Bulgaria. Antibiotics (Basel). 2021;10:940.
- [CrossRef] [PubMed] [Google Scholar]
- The characteristic of virulence, biofilm and antibiotic resistance of Klebsiella pneumoniae. Int J Environ Res Public Health. 2020;17:6278.
- [CrossRef] [PubMed] [Google Scholar]
- Biofilm of Candida albicans: Formation, regulation and resistance. J Appl Microbiol. 2021;131:11-22.
- [CrossRef] [PubMed] [Google Scholar]
- Analysis of the occurrence of antibiotic resistant bacteria in the hospital's effluent and its receiving environment. Microbiol Insights. 2022;15:11786361221078211.
- [CrossRef] [PubMed] [Google Scholar]
- Available from: https://www.cdc.gov/drugresistance/environment.html [Last accessed on 2022 Dec 14]
- Potential environmental and human health risks caused by antibiotic-resistant bacteria (ARB), antibiotic resistance genes (ARGs) and emerging contaminants (ECs) from municipal solid waste (MSW) landfill. Antibiotics (Basel). 2021;10:374.
- [CrossRef] [PubMed] [Google Scholar]
- Antimicrobial use and resistance in plant agriculture: A one health perspective. Agriculture. 2022;12:289.
- [CrossRef] [Google Scholar]
- Mechanisms of triazole resistance in Aspergillus fumigatus. Environ Microbiol. 2020;22:4934-52.
- [CrossRef] [PubMed] [Google Scholar]
- Herbicide selection promotes antibiotic resistance in soil microbiomes. Mol Biol Evol. 2021;38:2337-50.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotic resistance in aquaculture and aquatic organisms: A review of current nanotechnology applications for sustainable management. Environ Sci Pollut Res Int. 2022;29:69241-74.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotic-resistant bacteria in aquaculture and climate change: A challenge for health in the Mediterranean area. Int J Environ Res Public Health. 2021;18:5723.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotic resistance in agriculture: Perspectives on upcoming strategies to overcome upsurge in resistance. Curr Res Microb Sci. 2021;2:100030.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotic use in agriculture and its consequential resistance in environmental sources: Potential public health implications. Molecules. 2018;23:795.
- [CrossRef] [PubMed] [Google Scholar]
- Resistance genes, plasmids, multilocus sequence typing (MLST), and phenotypic resistance of non-typhoidal Salmonella (NTS) isolated from slaughtered chickens in Burkina Faso. Antibiotics (Basel). 2022;11:782.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotic resistance in agricultural soil and crops associated to the application of cow manure-derived amendments from conventional and organic livestock farms. Front Vet Sci. 2021;8:633858.
- [CrossRef] [PubMed] [Google Scholar]
- Antibiotics and antibiotic resistance genes in agricultural soils: A systematic analysis. Crit Rev Environ Sci Technol. 2022;53:1-18.
- [CrossRef] [Google Scholar]
- Easy access to antibiotics; Spread of antimicrobial resistance and implementation of one health approach in India. J Epidemiol Glob Health. 2021;11:444-52.
- [CrossRef] [PubMed] [Google Scholar]
- CDC establishes global networks to combat antimicrobial resistance. JAMA. 2022;327:315.
- [CrossRef] [Google Scholar]
- Futuristic non-antibiotic therapies to combat antibiotic resistance: A review. Front Microbiol. 2021;12:609459.
- [CrossRef] [PubMed] [Google Scholar]
- Effective antimicrobial combination in vivo treatment predicted with microcalorimetry screening. J Antimicrob Chemother. 2021;76:1001-9.
- [CrossRef] [PubMed] [Google Scholar]
- Synergistic effect of clinically available beta-lactamase inhibitors combined with cefiderocol against carbapenemase-producing gram-negative organisms. Antibiotics. 2022;11:1681.
- [CrossRef] [PubMed] [Google Scholar]
- β-lactamase inhibitors enhance the synergy between β-lactam antibiotics and daptomycin against methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 2016;61:e01564-16.
- [CrossRef] [PubMed] [Google Scholar]
- The synergistic antifungal effect and potential mechanism of D-penicillamine combined with fluconazole against Candida albicans. Front Microbiol. 2019;10:2853.
- [CrossRef] [PubMed] [Google Scholar]
- How CRISPR-Cas system could be used to combat antimicrobial resistance. Infect Drug Resist. 2020;13:1111-21.
- [CrossRef] [PubMed] [Google Scholar]
- CRISPR-Cas system: A potential alternative tool to cope antibiotic resistance. Antimicrob Resist Infect Control. 2020;9:131.
- [CrossRef] [PubMed] [Google Scholar]
- Conjugative delivery of CRISPR-Cas9 for the selective depletion of antibiotic-resistant enterococci. Antimicrob Agents Chemother. 2019;63:e01454-19.
- [CrossRef] [PubMed] [Google Scholar]
- Exploiting a conjugative CRISPR/Cas9 system to eliminate plasmid harbouring the mcr-1 gene from Escherichia coli. Int J Antimicrob Agents. 2019;53:1-8.
- [CrossRef] [PubMed] [Google Scholar]
- Nanomaterial-based therapeutics for antibiotic-resistant bacterial infections. Nat Rev Microbiol. 2021;19:23-36.
- [CrossRef] [PubMed] [Google Scholar]
- Nano-strategies to fight multidrug resistant bacteria-a battle of the titans. Front Microbiol. 2018;9:1441.
- [CrossRef] [PubMed] [Google Scholar]
- Nonviral genome editing based on a polymer-derivatized CRISPR nanocomplex for targeting bacterial pathogens and antibiotic resistance. Bioconjug Chem. 2017;28:957-67.
- [CrossRef] [PubMed] [Google Scholar]
- Silver nanoparticles and their antibacterial applications. Int J Mol Sci. 2021;22:7202.
- [CrossRef] [PubMed] [Google Scholar]
- Bacteriophage therapy: Clinical trials and regulatory hurdles. Front Cell Infect Microbiol. 2018;8:376.
- [CrossRef] [PubMed] [Google Scholar]
- Isolation and characterization of a potentially novel Siphoviridae phage (vB_SsapS-104) with lytic activity against Staphylococcus saprophyticus isolated from urinary tract infection. Folia Microbiol (Praha). 2019;64:283-94.
- [CrossRef] [PubMed] [Google Scholar]
- Characterization of a lytic bacteriophage vB_EfaS_PHB08 harboring endolysin Lys08 against Enterococcus faecalis biofilms. Microorganisms. 2020;8:1332.
- [CrossRef] [PubMed] [Google Scholar]
- Synergy between the host immune system and bacteriophage is essential for successful phage therapy against an acute respiratory pathogen. Cell Host Microbe. 2017;22:38-47.e4.
- [CrossRef] [PubMed] [Google Scholar]
- The role of vaccines in combatting antimicrobial resistance. Nat Rev Microbiol. 2021;19:287-302.
- [CrossRef] [PubMed] [Google Scholar]
- Predicting the impact of typhoid conjugate vaccines on antimicrobial resistance. Clin Infect Dis. 2019;68(Suppl 2):S96-104.
- [CrossRef] [PubMed] [Google Scholar]
- Available from: https://www.emro.who.int/pak/pakistan-news/pakistan-first-country-to-introduce-new-typhoid-vaccine-into-routine-immunization-programme.html [Last accessed on 2022 Dec 17]
- The inverse relationship between influenza vaccination and antimicrobial resistance: An ecological analysis of Italian data. Vaccines (Basel). 2022;10:554.
- [CrossRef] [PubMed] [Google Scholar]
- The role of vaccines in preventing bacterial antimicrobial resistance. Nat Med. 2018;24:10-9.
- [CrossRef] [PubMed] [Google Scholar]
- Antimicrobial peptides: Classification, design, application and research progress in multiple fields. Front Microbiol. 2020;11:582779.
- [CrossRef] [PubMed] [Google Scholar]
- Antimicrobial peptides: Mechanism of action, activity and clinical potential. Mil Med Res. 2021;8:48.
- [CrossRef] [PubMed] [Google Scholar]
- Pretreatment with cathelicidin-BF ameliorates Pseudomonas aeruginosa pneumonia in mice by enhancing NETosis and the autophagy of recruited neutrophils and macrophages. Int Immunopharmacol. 2018;65:382-91.
- [CrossRef] [PubMed] [Google Scholar]
- Antimicrobial peptides: A potent alternative to antibiotics. Antibiotics (Basel). 2021;10:1095.
- [CrossRef] [PubMed] [Google Scholar]
- Corrigendum: The role of fecal microbiota transplantation in reducing intestinal colonization with antibiotic-resistant organisms: The current landscape and future directions. Open Forum Infect Dis. 2019;6:ofz391.
- [CrossRef] [PubMed] [Google Scholar]
- Dysbiosis of gut microbiota in patients undergoing cardiac surgery. Glob J Med Pharm Biomed Update. 2022;17:13.
- [CrossRef] [Google Scholar]
- 1805 use of stool transplant to clear fecal colonization with carbapenem-resistant Enterobacteraciae (CRE): Proof of concept. Open Forum Infect Dis. 2014;1(Suppl 1):S65.
- [CrossRef] [Google Scholar]
- Fecal microbial transplantation for the treatment of persistent multidrug-resistant Klebsiella pneumoniae infection in a critically ill patient. Case Rep Infect Dis. 2020;2020:8462659.
- [CrossRef] [PubMed] [Google Scholar]
- Fecal microbiota transplantation in patients with blood disorders inhibits gut colonization with antibiotic-resistant bacteria: Results of a prospective, single-center study. Clin Infect Dis. 2017;65:364-70.
- [CrossRef] [PubMed] [Google Scholar]
- Mechanisms of action of probiotics. Adv Nutr. 2019;10(Suppl 1):S49-66.
- [CrossRef] [PubMed] [Google Scholar]
- Probiotics, prebiotics and synbiotics-a review. J Food Sci Technol. 2015;52:7577-87.
- [CrossRef] [PubMed] [Google Scholar]
- In vitro antibacterial effect of probiotics against carbapenamase-producing multidrug-resistant Klebsiella pneumoniae clinical isolates, Cairo, Egypt. J Egypt Public Health Assoc. 2022;97:19.
- [CrossRef] [PubMed] [Google Scholar]
- A new use of Lactobacillus rhamnosus GG administration in the NICU: Colonized vancomycin-resistant Enterococcus eradication in the gastrointestinal system. J Matern Fetal Neonatal Med. 2022;35:1192-8.
- [CrossRef] [PubMed] [Google Scholar]
- Policy document on antimicrobial stewardship practices in India. Indian J Med Res. 2019;149:180-4.
- [CrossRef] [PubMed] [Google Scholar]
- WHO Competency Framework for Health Workers' Education and Training on Antimicrobial Resistance Geneva: World Health Organization; 2018.
- [Google Scholar]
- Available from: https://reliefweb.int/report/world/global-antimicrobial-resistance-and-use-surveillance-system-glass-report-2022 [Last accessed on 2022 Dec 18]
- Feasibility, challenges, and benefits of global antimicrobial resistance surveillance system implementation: Results from a multicenter quasi-experimental study. Antibiotics (Basel). 2022;11:348.
- [CrossRef] [PubMed] [Google Scholar]
- Educational activities for students and citizens supporting the one-health approach on antimicrobial resistance. Antibiotics (Basel). 2021;10:1519.
- [CrossRef] [PubMed] [Google Scholar]







